Electron Energy Loss Spectroscopy (EELS)
Home » Our Techniques » Spectroscopy » EELS
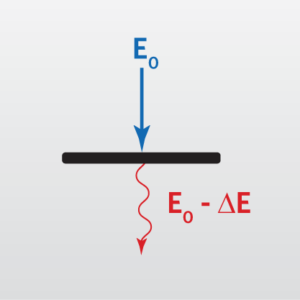
Electron Energy Loss Spectroscopy (EELS) is a technique that provides elemental information on the nanometer scale when coupled with Transmission Electron Microscopy (TEM) and Scanning Transmission Electron Microscopy (STEM). The energy of the incident electrons is altered (reduced) as they pass through the sample. This energy loss can be characterized using EELS to provide elemental identification. Compared to Energy Dispersive X-ray Spectroscopy (EDS), EELS provides improved signal to noise, better spatial resolution (down to 1 nm), higher energy resolution (<1 eV for EELS, 0.1-0.2eV for monochromated EELS) and increased sensitivity to the lower atomic number elements. For some elements, chemical bonding information can be obtained. In addition, more information such as bandgap, dielectric function, refractive index can be extracted by monochromated EELS.
Ideal Uses of EELS
- Elemental identification and mapping
- Elemental identification (spot analysis, linescans, 2D chemical maps)
- Chemical fingerprinting (limited cases)
- Bandgap
- Dielectric function, refractive index
Strengths
- More signal collection than EDS
- Especially useful in the Si/C/O/N system
- ~0.2nm probe size higher energy resolution, can sometimes provide chemical information
- Higher sensitivity to Low Z elements
Limitations
- Longer to set up
- Multi element detection sometimes requires multiple set ups
- Backgrounds and peak shapes are complicated
- Good detection of High Z elements can be problematic
EELS Technical Specifications
- Elements Detected: Li-U
- Detection Limits: 0.1-0.5%
- Lateral Resolution/Probe Size: 1 nm (atomic level is possible for certain materials)
Would you like to learn more about using Electron Energy Loss Spectroscopy?
Contact us today for your Electron Energy Loss Spectroscopy (EELS) needs. Please complete the form below to have an EAG expert contact you.